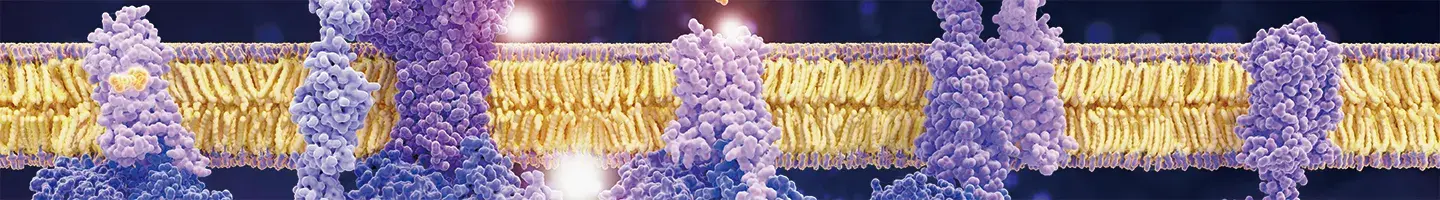
|
5-hydroxytryptamine
|
5-hydroxytryptamine Receptor 1A (HTR1A) |
|
5-hydroxytryptamine
|
5-hydroxytryptamine Receptor 1E (HTR1E) |
|
5-hydroxytryptamine
|
5-hydroxytryptamine Receptor 2C (HTR2C) |
|
5-hydroxytryptamine
|
5-hydroxytryptamine Receptor 4 (HTR4) |
|
5-hydroxytryptamine
|
5-hydroxytryptamine Receptor 5A (HTR5A)
|
|
5-hydroxytryptamine
|
5-hydroxytryptamine Receptor 7 (HTR7) |
|
Acetylcholine
|
Cholinergic Receptor Muscarinic 1 (mAChR M1) |
|
Acetylcholine
|
Cholinergic Receptor Muscarinic 3 (mAChR M3) |
|
Adenosine
|
Adenosine A1 Receptor (ADORA1) |
|
Adenosine
|
Adenosine A2a Receptor (ADORA2A) |
| Adenosine |
Adenosine A2b Receptor (ADORA2B)
|
|
Adrenoceptors
|
Alpha 1a Adrenergic Receptor (ADRA1A)
|
|
Adrenoceptors
|
Alpha 1b Adrenergic Receptor (ADRA1B) |
| Adrenoceptors |
Alpha 1d Adrenergic Receptor (ADRA1D) |
| Adrenoceptors |
Alpha 2a Adrenergic Receptor (ADRA2A) |
|
Adrenoceptors
|
Alpha 2b Adrenergic Receptor (ADRA2B) |
| Adrenoceptors |
Beta 1 Adrenergic Receptor (ADRB1) |
|
Adrenoceptors
|
Beta 2 Adrenergic Receptor (ADRB2)
|
|
Adrenoceptors
|
Beta 3 Adrenergic Receptor (ADRB3) |
| Angiotensin |
Angiotensin II Receptor Type 1 (AGTR1) |
| Apelin |
Apelin Receptor (APLNR) |
|
Bradykinin
|
Bradykinin Receptor B2 (BDKRB2) |
|
Cannabinoid
|
Cannabinoid receptor 1 (CNR1) |
| Cannabinoid |
Cannabinoid Receptor 2 (CNR2) |
| Chemokine |
C-C Motif Chemokine Receptor 1 (CCR1) |
| Chemokine |
C-C Motif Chemokine Receptor 2 (CCR2) |
| Chemokine |
C-C Motif Chemokine Receptor 4 (CCR4) |
| Chemokine |
C-C Motif Chemokine Receptor 5 (CCR5) |
| Chemokine |
C-C Motif Chemokine Receptor 7 (CCR7) |
| Chemokine |
C-C Motif Chemokine Receptor 9 (CCR9) |
| Chemokine |
C-C Motif Chemokine Receptor 10 (CCR10) |
| Chemokine |
Atypical Chemokine Receptor 4 (CCR11) |
|
Chemokine
|
C-C motif chemokine receptor like 2 (CRL2) |
| Chemokine |
C-X-C Motif Chemokine Receptor 1 (CXCR1) |
| Chemokine |
C-X-C Motif Chemokine Receptor 2 (CXCR2) |
| Chemokine |
C-X-C Motif Chemokine Receptor 3 (CXCR3) |
| Chemokine |
C-X-C Motif Chemokine Receptor 4 (CXCR4) |
| Chemokine |
C-X-C Motif Chemokine Receptor 5 (CXCR5) |
| Chemokine |
C-X-C Motif Chemokine Receptor 6 (CXCR6) |
| Chemokine |
Atypical Chemokine Receptor 3 (CXCR7) |
|
Chemokine
|
C-X3-C Motif Chemokine Receptor 1 (CX3CR1) |
|
Chemokine
|
Chemokine Receptor D6 (ACKR2) |
|
Cholecystokinin
|
Cholecystokinin B Receptor (CCKBR) |
|
Class A Orphans
|
G Protein-Coupled Receptor 3 (GPR3) |
| Class A Orphans |
G Protein-Coupled Receptor 15 (GPR15)
|
| Class A Orphans |
G Protein-Coupled Receptor 21 (GPR21)
|
|
Class A Orphans
|
G Protein-Coupled Receptor 23 (GPR23) |
|
Class A Orphans
|
G Protein-Coupled Receptor 31 (GPR31)
|
|
Class A Orphans
|
G Protein-Coupled Receptor 37 (GPR37)
|
|
Class A Orphans
|
G Protein-Coupled Receptor 37 Like 1 (GPR37L1) |
|
Class A Orphans
|
G Protein-Coupled Receptor 45 (GPR45) |
| Class A Orphans |
G Protein-Coupled Receptor 50 (GPR50)
|
|
Class A Orphans
|
G Protein-Coupled Receptor 52 (GPR52) |
|
Class A Orphans
|
G Protein-Coupled Receptor 62 (GPR62) |
|
Class A Orphans
|
G Protein-Coupled Receptor 83 (GPR83)
|
|
Class A Orphans
|
G Protein-Coupled Receptor 84 (GPR84)
|
|
Class A Orphans
|
G Protein-Coupled Receptor 88 (GPR88) |
|
Class A Orphans
|
G Protein-Coupled Receptor 126 (GPR126) |
|
Class A Orphans
|
G Protein-Coupled Receptor 132 (GPR132) |
|
Class A Orphans
|
G Protein-Coupled Receptor 135 (GPR135) |
|
Class A Orphans
|
G Protein-Coupled Receptor 137 (GPR137) |
|
Class A Orphans
|
G Protein-Coupled Receptor 139 (GPR139)
|
|
Class A Orphans
|
G Protein-Coupled Receptor 146 (GPR146) |
| Class A Orphans |
G Protein-Coupled Receptor 148 (GPR148) |
|
Class A Orphans
|
G Protein-Coupled Receptor 150 (GPR150) |
|
Class A Orphans
|
G Protein-Coupled Receptor 151 (GPR151) |
|
Class A Orphans
|
G Protein-Coupled Receptor 152 (GPR152)
|
|
Class A Orphans
|
G Protein-Coupled Receptor 153 (GPR153) |
|
Class A Orphans
|
G protein-Coupled Receptor 157 (GPR157) |
|
Class A Orphans
|
Transmembrane Protein Adipocyte Associated 1 (GPR175) |
| Class A Orphans |
G Protein-Coupled Receptor 176 (GPR176)
|
| Class A Orphans |
G protein-Coupled Receptor 182 (GPR182)
|
| Class A Orphans |
Leucine Rich Repeat Containing G Protein-Coupled Receptor 5 (LGR5) |
| Class A Orphans |
Leucine Rich Repeat Containing G Protein-Coupled Receptor 6 (LGR6) |
| Class A Orphans |
MAS Related GPR Family Member F (MRGPRF)
|
|
Class A Orphans
|
MAS Related GPR Family Member G (MRGPRG) |
|
Class A Orphans
|
MAS Related GPR Family Member X4 (MRGPRX4) |
| Class A Orphans |
MAS related GPR family member X1 (MRGX1) |
| Complement |
Complement C3A Receptor 1 (C3AR1) |
|
Complement
|
Complement Component 5a Receptor 2 (C5L2) |
| Dopamine |
Dopamine Receptor D1 (DRD1) |
| Dopamine |
Dopamine Receptor D2 (DRD2) |
| Dopamine |
Dopamine Receptor D4 (DRD4) |
| Dopamine |
Dopamine Receptor D5 (DRD5) |
| Endothelin |
Endothelin Receptor Type B (Ednrb) |
|
Estrogen
|
G protein-coupled estrogen receptor 1 (GPR30) |
|
Formylpeptide
|
Formyl Peptide Receptor 1 (FPR1)
|
| Formylpeptide |
Formyl Peptide Receptor 2 (FPRL1) |
|
Free fatty acid
|
Free Fatty Acid Receptor 1 (GPR40)
|
| Free fatty acid |
Free Fatty Acid Receptor 2 (GPR43) |
| Free fatty acid |
Free Fatty Acid Receptor 4 (GPR120) |
|
Ghrelin
|
Growth Hormone Secretagogue Receptor (GHS-R1) |
|
Glycoprotein
|
Follicle Stimulating Hormone Receptor (FSHR) |
|
Glycoprotein
|
Luteinizing Hormone/choriogonadotropin Receptor (LHR) |
|
Glycoprotein
|
Thyroid Stimulating Hormone Receptor (TSHR)
|
|
Gonadotrophin
|
Gonadotropin Releasing Hormone Receptor (GnRHR) |
| Histamine |
Histamine Receptor H1 (HRH1) |
|
Hydroxycarboxylic acid
|
Hydroxycarboxylic Acid Receptor 1 (GPR81) |
|
Kisspeptin
|
KISS1 Receptor (GPR54) |
|
Leukotriene
|
Leukotriene B4 Receptor 2 (LTB4R2) |
|
Leukotriene
|
Oxoeicosanoid Receptor 1 (OXER1) |
|
Lysophospholipid
|
Lysophosphatidic Acid Receptor 2 (EDG-4/LPAR2) |
| Lysophospholipid |
PAF Receptor (PTAFR) |
| Lysophospholipid |
Sphingosine-1-phosphate Receptor 2 (S1PR2) |
|
Lysophospholipid
|
Sphingosine-1-phosphate Receptor 4 (S1PR4) |
| Lysophospholipid |
Sphingosine-1-phosphate Receptor 5 (S1PR5) |
| Melanin-concentrating hormone |
Melanin Concentrating Hormone Receptor 1 (MCH-1R)
|
| Melanocortin |
Melanocortin 1 Receptor (MC1R) |
| Melanocortin |
Melanocortin 4 Receptor (MC4R) |
| Melanocortin |
Melanocortin 5 Receptor (MC5R) |
|
Melatonin
|
Melatonin Receptor 1A (MTNR1A) |
|
Motilin
|
Motilin Receptor (MLNR) |
|
Neuromedin U
|
Neuromedin U Receptor 2 (NMUR2)
|
|
Neuropeptide W/ B
|
Neuropeptides B and W Receptor 1 (GPR7)
|
|
Neuropeptide W/ B
|
Neuropeptides B and W Receptor 2 (GPR8)
|
|
Neurotensin
|
Neurotensin Receptor 2 (NTSR2) |
|
Olfactory Receptor
|
Olfactory Receptor Family 2 Subfamily W Member 3 (OR2W3) |
| Opioid |
Opioid Receptor Delta 1 (OPRD1) |
| Opioid |
Opioid Receptor Mu 1 (OPRM1) |
|
Opsins
|
Opsin 5 (OPN5)
|
|
Opsins
|
Opsin Blue (OPN1SW)
|
| Opsins |
Opsin Red / Green (OPN1MW2) |
| Opsins |
Retinal G Protein Coupled Receptor (RGR) |
| Opsins |
Rhodopsin (RHO) |
|
Orexin
|
Orexin Receptor 2 (OX2R) |
|
Prolactin
|
Prolactin Releasing Hormone Receptor (GPR10)
|
| Prostanoid |
Prostaglandin E Receptor 3 (PTGER3) |
|
Prostanoid
|
Thromboxane A2 Receptor (TXA2R) |
|
Proteinase-activated receptor
|
F2R Like Trypsin Receptor 1 (PAR2)
|
|
Proteinase-activated receptor
|
Coagulation Factor II Thrombin Receptor Like 2 (PAR3) |
|
Relaxin family
|
Relaxin Family Peptide Receptor 1 (LGR7) |
|
Relaxin family
|
Relaxin Family Peptide Receptor 3 (RXFP3) |
| P2Y |
Purinergic Receptor P2Y1 (P2Y1) |
| P2Y |
Purinergic Receptor P2Y2 (P2Y2) |
| P2Y |
Purinergic Receptor P2Y4 (P2Y4)
|
| P2Y |
Purinergic Receptor P2Y11 (P2Y11) |
| P2Y |
Purinergic Receptor P2Y12 (P2Y12) |
| Somatostatin |
Somatostatin Receptor 2 (SSTR2) |
| Somatostatin |
Somatostatin Receptor 3 (SSTR3) |
| Somatostatin |
Somatostatin Receptor 4 (SSTR4) |
| Somatostatin |
Somatostatin Receptor 5 (SSTR5) |
| Tachykinin |
Neurokinin 1 Receptor (NK1R) |
|
Tachykinin
|
Neurokinin 2 Receptor (TACR2) |
|
Tachykinin
|
Neurokinin 3 Receptor (TACR3) |
|
Urotensin
|
Urotensin 2 Receptor (GPR14)
|
| Vasopressin |
Arginine Vasopressin Receptor 1B (AVPR1B) |